
How Old is the Earth?
|
|
According to National Geographic:
 |
 |
|
“The
world’s oldest known rock was dated . . . at 4.03 billion years.
The earth itself goes back 4.5 billion years.”
(National
Geographic, September 2001, p. 91)
|
To those who believe in secular evolution, an old earth is considered essential -
– “The secrets of evolution are death and time - the death of enormous numbers of life forms that were imperfectly adapted to the environment; and time for a long succession of small mutations that were by accident adaptive, time for the slow accumulation of patterns of favorable mutations [emphasis added].” (Carl Sagan , Cosmos, 1980, p.30)
– Time is in fact the hero of the plot . . . given so much time the ‘impossible’ becomes possible, the possible probable and the probable virtually certain. One has only to wait: time itself performs miracles [emphasis added]. (George Wald, “The Origin of Life,” Physics and the Chemistry of Life, 1955, p.12)
 |
 |
The Bible teaches that the earth is about 6000 years old
If we recognize that the “the heavens and the earth and all that is in them” were created in six ordinary days and we interpret the genealogies in a straightforward, literal manner then we have to conclude that the earth is about 6000 years old:
| From Creation to the Abraham: | 2,000 years | |
| From Abraham to Christ: | 2,000 years | |
| From Christ to the Present: | 2,000 years | |
| Age of the Human Race: |
6,000 years |
Or as Hebrew professor James Barr put it:
“Probably, so far as I know, there is no professor of Hebrew or Old Testament at any world-class university who does not believe that the writer(s) of Genesis 1-11 intended to convey to their readers the ideas that
(a) Creation took place in a series of six days which were the same as the days of 24 hours we now experience
(b) The figures contained in the Genesis genealogies provided by simple addition a chronology from the beginning of the world up to later stages in the biblical story . . .
Or, to put it negatively, the apologetic arguments which suppose the 'days' of creation to be long eras of time [and] the figures of years not to be chronological . . . are not taken seriously by any such professors, as far as I know.” [emphasis added]
(James Barr in a letter to David Watson, 1984. Barr is Professor of Hebrew Bible, Vanderbilt University, and former Reguis Professor of Hebrew, Oxford University, Oxford, England.)
How Can We Know How Old Something Is?
 |
A. First Hand Knowledge - We were there personally to observe the creation taking place.
B. A Reliable Eyewitness - We have received a reliable eyewitness account telling us when the item in question was made from someone who was there to see the creation taking place.
C. A Reliable "Clock" - We have the current reading from a reliable "clock" that we are sure has been running since the time the object was created. And in order for us to determine the age of the item in question from this "clock" we need to know three things about the clock:
– The Original Setting: What the clock read at the time that the item in question was created.
– Runs at a Known Rate - That the clock has been running at a constant rate (or if the rate has changed, we must know exactly how it changed and when)
– Has Never Been Changed - That the clock has never been changed or reset since the creation of the item in question (or if the clock has been changed, we must know exactly how it changed and when)
Concerning the Age of the Earth:
– Option A - We weren’t there (Job 38:4).
– Option B - Those who believe that the Bible is the inerrant word of the living God and believe that Genesis 1 teaches the earth was made in six days and the genealogies are reliable - believe that they have option “B”: a reliable eyewitness account.
– Option C - Those who do not accept Genesis as a reliable account of when God created the earth (either because they think that Genesis is not reliable on this point or they read indefinite periods time into the text of Genesis 1) must rely on option “C”. The “clock” most often chosen by secular scientists by which they arrive at such ancient dates is the “clock” of radioactive decay.
Radioactive Dating Methods
 |
• The most common dating methods used by scientists today are the radioactive dating methods.
• How radioactive dating works:
– Radioactive elements (known as the "parent" element) give off radiation at an (assumed) constant rate.
– As this happens, the radioactive element turns into another (usually non-radioactive) element (known as the "daughter" element).
– The time it takes for half of any parent element to turn into the daughter element is called the "half-life" of that element.
– By measuring the amount of parent and daughter elements present in a rock, scientists (given certain assumptions) believe that they can determine the age of that rock.
Some examples of radioactive elements used in dating rocks are:
|
Parent-Daughter |
Half Life |
|
uranium-lead |
4.5 billion years |
|
potassium-argon |
1.3 billion years |
|
rubidium-strontium |
4.7 billion years |
• Note: These radioactive dating methods can only be used on:
– Igneous Rocks (formed by volcanic action)
– Or Metamorphic Rocks (igneous rocks that have undergone change).
– Not Sedimentary Rocks (rocks laid down as sediment by water).
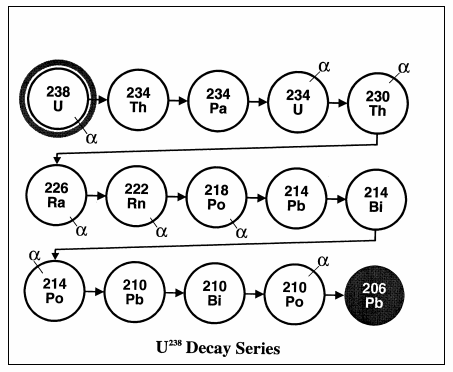
Note: for simplicity's sake we describe the deterioration from a parent element (e.g. uranium) to a daughter element (e.g. lead). The deterioration process is actually a little more complex than that. Note, for example, the stages that Uranium goes through in deteriorating into lead:
 |
Apparent Problems with Radioactive Dating
• Scientists often encounter examples in the real world that contradict their assumptions about the reliability of radioactive dating methods and the presumption of long geological ages for the earth:
– Scientists often get conflicting dates using different radioactive dating methods on the same rock. Obviously both dates can't be right!
– Sometimes rocks that are thought to be much older than other rocks (because they are buried several layers under them) date much younger than the rocks found above them!
– Often rocks that are known to be very young (because they come from recent volcanoes whose eruption date is known) give dates using radioactive dating methods that are very old and therefore known to be erroneous
|
Volcano |
Actual Age of Rocks Formed |
Age Given by Radioactive Dating |
|
Sunset Crater, Arizona |
900 years |
210,000 to 230,000 years |
|
Mt. Rangitoto, New Zealand |
300 years |
485,000 years |
|
Hualalai Volcano, Hawaii |
200 years |
140 million to 2.96 billion years |
Above examples are documented by John Morris, Ph.D., The Young Earth, 1994, p.54-55
• It is obvious that radiometric methods may not be the reliable dating methods they are often claimed to be. Age estimates on a given geological stratum using different methods are often quite different (sometimes by hundreds of millions of years). There is no absolutely reliable long-term radiological clock
– William D. Stansfield, The Science of Evolution, New York: Macmillan, 1977, p.84
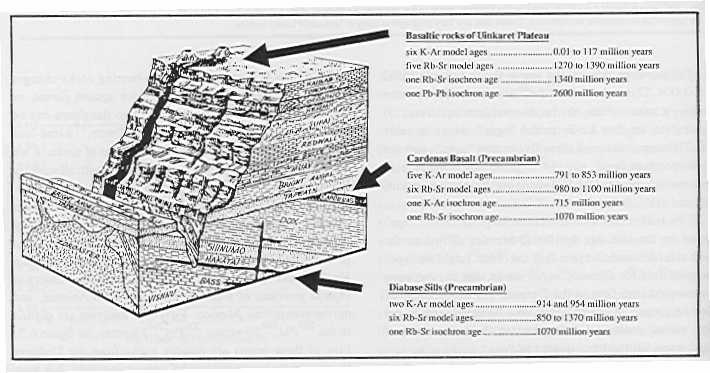
“Ages” of Grand Canyon Rocks
 |
|
As documented by Steve Austin Ph.D., Grand Canyon - Monument to Catastrophe, 1994, p.126 |
Note the vast discrepancies in ages resulting from different dating methods!
More Examples of Radioactive Dating Problems
Example#1
In 1993, several pieces of wood were found entombed in the bottom of a (cooled) lava flow in a coal mine in Australia. Parts of the wood were still intact! The various pieces of wood were carbon dated between 35,000 - 45,000 years old. The lava enclosing the wood was dated (using K-Ar dating) at 36.7 ± 1.2 million years old!
 |
 |
Dr. Andrew Snelling, Creation ex nihilo, December 1997 - February 1998, Vol. 20 No. 1, p.24-27
Example#2
Rock from cooled lava flows known to have occurred between 1949 and 1975 were sent to several standard labs to be dated - without telling the labs where the rocks had come from or that the actual age of the rocks was already known. The labs (using K-Ar) dating returned results ranging from <0.27 million years old to 3.5 ± .2 million years old!
 |
 |
Dr. Andrew Snelling, Creation ex nihilo, December 1999 - February 2000, Vol. 22 No. 1, p.18-21
Example#3
Rocks from the cooled lava dome of Mount St. Helens (which at the time were about 10 years old) were sent to a reputable lab to be dated - without telling the lab where the rocks had come from. The lab (using K-Ar) dating returned results ranging from
0.34 ± 0.06 million years old to
2.8 ± 0.6 million years old!
 |
 |
Keith Swensen, Creation, June - August 2001, Vol. 23 No. 3, p.23-25
The Dating Game
 |
A great illustration of the games evolutionists play when dating a particular rock or fossil can be seen in a sequence of events described by Marvin Lubenow in the appendix of his book, Bones of Contention, 1992
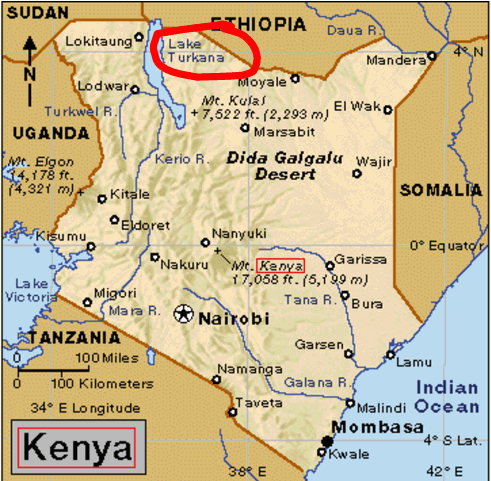 |
Background: In 1972, Richard Leakey discovered a very modern looking skull in Kenya just east of Lake Turkana. At the time, fossils found in this area were considered about 2.9 million years old. Leakey’s discovery created a problem, because the theory of human evolution did not allow for a skull that looked so modern to be so old! The following chart gives a history of how the evolutionists ended up “solving” this problem. Notice how many different dates evolutionists can get for the same layer of rock!
Year |
Dated by Whom |
Dating Method |
Age Claimed |
Comment |
|
1969 |
Fitch and Miller |
potassium-argon |
212 to 230 (million years old) |
Original Dating of the rocks above the area where Leakey later found his skull. |
|
1970 |
Fitch and Miller |
potassium-argon argon-argon
|
2.61 ± .26 |
The original date conflicted with the dating of fossils in the area so they asked for new samples |
|
1972 |
|
|
2.9 |
Richard Leakey discovered a very modern looking skull. |
|
1972 |
Vincent Maglio |
fossils of pigs and elephants found in the area |
2.61 |
Confirmed Fitch and Miller’s date |
|
1974 |
Brock and Isaac |
paleomagnetism |
2.7 to 3.0 |
Based on samples below area tested by Fitch and Miller, thus confirming their dates |
|
1974 |
Fitch and Miller |
|
2.61 ± .26 |
Published their revised study |
|
1975 |
Basil Cooke |
pig fossils in the area |
1.8 |
Presented at a geology conference in London |
|
1975 |
G. H. Curtis et al. |
potassium-argon |
1.82 |
They challenged the validity of Fitch and Miller’s methodology |
|
1976 |
Fitch and Miller |
potassium-argon argon-argon |
2.4 |
Re-tested both sets of rocks using more advanced techniques. |
|
1976 |
Hurford et al. |
fission-track dating |
2.44 |
|
|
1977 |
G.A. Wagner |
fission-track dating |
1.9 |
Claimed that Hurford et al. used the wrong radiactive constant in doing their calculations |
|
1980 |
Gleadow et al. |
fission-track dating and potassium-argon |
1.87 or 1.89 |
|
|
1981 |
Ian McDougal |
Argon-argon |
1.88 |
Published in Nature magazine |
|
Today |
|
|
1.9 |
Leakey’s skull now fits the traditional evolutionary view |
The Dating Game - Conclusion
“The pigs won. In the ten-year controversy over the dating of one of the most important human fossils ever discovered, the pigs won. The pigs won over the elephants. The pigs won over K-Ar dating. The pigs won over Ar-Ar dating. The pigs won over fission-track dating. They won over paleomagnetism. The pigs took it all. But in reality, it wasn't the pigs that won. It was evolution that won. In the dating game, evolution always wins.” (Lubenow, p.266)
Carbon-14 Dating
 |
Some Basic Facts About Carbon Dating:
• Carbon-14 dating can only be used on organic materials, not on rocks or even fossils
• The half-life of C-14 is about 5,730 years
• Scientists will usually not use Carbon-14 dating on anything that they consider to be older than 50,000 years
What is Carbon-14?
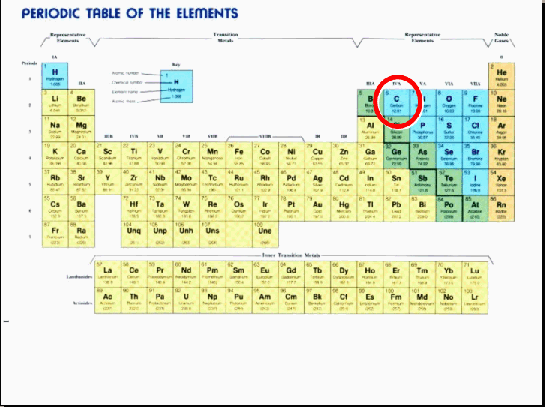
• A normal carbon atom weighs 12 atomic mass units.
 |
 |
• However, about one in every trillion carbon atoms (in earth’s atmosphere today) weighs 14 atomic units.
• This carbon is called carbon-14.
• It is also called radiocarbon because it is radioactive (but not dangerous).
• Half of all carbon-14 will decay in about 5,730 years (from the time it was formed) to form nitrogen.
• Half of the remainder will decay into nitrogen in another 5,730 years, and so on.
How Carbon-14 Dating Works
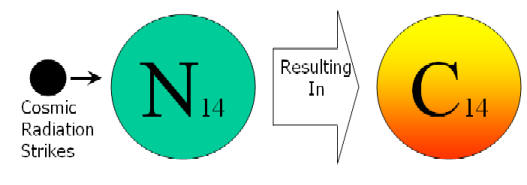
• When Nitrogen in the earth’s upper atmosphere is struck by cosmic radiation it turns into radioactive carbon (Carbon-14).
 |
• In this way, about 21 pounds of Nitrogen is converted each year into radiocarbon (carbon-14)
• Most carbon-14 quickly combines with oxygen to form radioactive carbon dioxide, which then intermingles with regular carbon dioxide throughout the earth’s atmosphere.
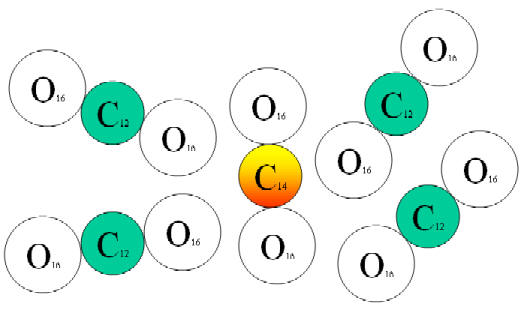 |
• Plants use carbon dioxide. In doing this, plants absorb small amounts of radioactive carbon into their cells.
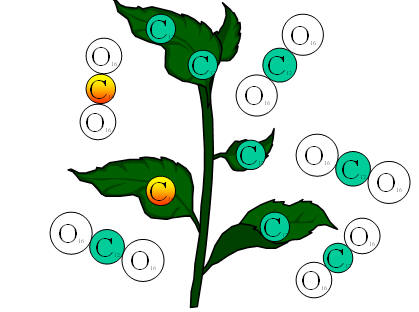 |
• Animals eat plants. In doing this, animals absorb small amounts of radioactive carbon into their bodies.
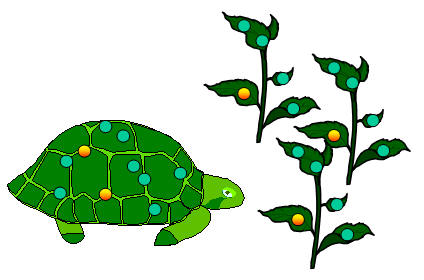 |
• When plants and animals die, they stop absorbing radioactive carbon.
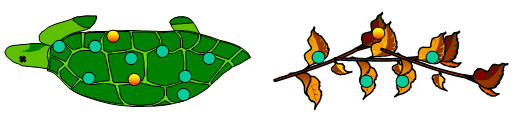 |
• The radioactive carbon in a plant or animal will slowly decay back into nitrogen after the plant or animal dies.
 |
• By measuring the amount of radioactive carbon left in a dead plant or animal (and by making certain assumptions) scientists believe that they can determine how long ago the plant or animal has died.
 |
Assumptions Made in Radiocarbon Dating
1. A constant rate of decay - i.e. the half-life of carbon-14 has always been 5,730 years
2. No loss or gain of parent or daughter - i.e. the specimen being dated has not taken in or lost any carbon-14 since the time it died (e.g through contamination).
3. Known amounts of daughter at the start. This requires that the following things be true:
– The ratio of carbon-14 to carbon-12 that exists in the earth’s atmosphere today has always been the same. Has it always been the same?
– The ratio of carbon-14 to carbon-12 that exists in all living things always matches the ratio that exists in the earth’s atmosphere. Does it?
Has the ratio of carbon-14 to carbon-12 that exists in the earth’s atmosphere always been the same as it is today?
• When the earth was first created, there would not have been any naturally formed carbon-14 in the atmosphere, since it takes time for carbon-14 to form.
• Over time, as carbon-14 began to form, the ratio of carbon-14 to carbon-12 would increase.
• It is generally assumed that, after about 30,000 years, the amount of carbon-14 disappearing due to radioactive decay will equal the amount of new carbon-14 being formed in the upper atmosphere. Thus the ratio of carbon-14 to carbon-12 will reach an equilibrium point.
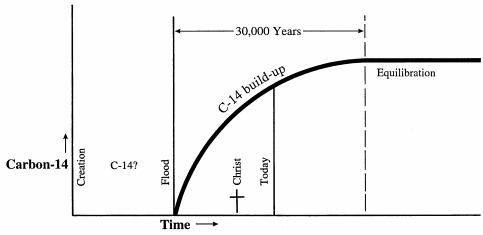 |
• Since Evolutionists assume that the earth is billions of years old, they assume that the formation of carbon-14 reached equilibrium years ago, and has therefore been constant for billions of years.
• Willard Libby, who received a Nobel Prize for developing carbon dating, conducted tests in 1950 and found that the carbon-14 to carbon 12 ratio had not reached equilibrium, but was still increasing! But he ignored the results, assuming that they had to be erroneous! After all, he “knew” the earth was more than 30,000 years old! (Walter Brown, In the Beginning, 7th Edition, p.244-246)
• In 1977, Melvin Cook did similar but more concise tests which showed that the ratio of carbon-14 to carbon-12 in the earth’s atmosphere was definitely increasing, even faster than Libby’s test indicated! (Walter Brown, In the Beginning, 7th Edition, p.244-246)
Problems with the Assumptions of Carbon-14 Dating in Light of Biblical Teaching:
• The Bible teaches that the earth was created about 6000 years ago, therefore the ratio of carbon-14 to carbon-12 has not reached equilibrium and is still increasing.
• The Bible teaches that about 4,500 years ago there was a global flood. Conditions on the earth before, during, and after the flood might have had a dramatic effect on the ratio of carbon-14 to carbon-12.
– If more lush forests and greater land area existed prior to the flood than exist today, it might have resulted in a lower ratio of carbon-14 to carbon-12 in the earth’s atmosphere.
– If the “springs of the great deep” which “burst forth” during the flood (Genesis 7:11) were under great pressure before they burst forth, they might have released large amounts of carbon dioxide into the earth’s atmosphere during the flood - diluting the carbon-14 thereby reducing the ratio of carbon-14 to carbon-12.
– If Genesis 1:6-7 teaches that there was a water canopy above the earth’s atmosphere prior to the flood, then nitrogen in the upper atmosphere may have been shielded from cosmic rays, which might mean that no carbon-14 was formed prior to the flood!
Is Carbon Dating Reliable?
• There is reason to believe that the ratio of carbon-14 to carbon-12 in the earth’s atmosphere might have been much lower thousands of years ago than it is today.
• If this is the case, then the ratio of carbon-14 to carbon-12 in living things might have also been much lower than it is in living things today (if we assume that the ratio of C-14 to C-12 in living things is the same as the ratio in the earth’s atmosphere).
• If this is the case, then living things that died thousands of years ago, will appear much older than they really are to an evolutionist who assumes that the ratio of C-14 to C-12 has always been the same.
– The evolutionist will assume that a low level of carbon-14 in a plant or animal that died thousands of years ago is exclusively due to all of the original carbon-14 having decayed over a period of many thousand years.
– In reality, the low level of carbon-14 in a plant or animal that died thousands of years ago may only be partially due to carbon-14 decay. The low level of carbon-14 may also be due to the fact that the original level of carbon-14 was low in the first place due to differing conditions on the earth at the time the plant or animal died.
Examples of Problems in Carbon Dating
o Living mollusk shells were carbon dated as being 2300 years old. (Science vol. 141, 1963 p. 634-637)
o A freshly killed seal was carbon dated as having died 1300 years ago! (Antarctic Journal vol. 6 Sept-Oct. 1971 p. 211)
 |
o Shells from living snails were carbon dated as being 27,000 years old. (Science Vol. 224, 1984 p. 58-61)
 |
o “One part of the Vollosovitch mammoth carbon dated at 29,500 years old and another part at 44,000.” (Troy L. Pewe, Quaternary Stratigraphic Nomenclature in Unglaciated Central Alaska, Geological Survey Professional Paper 862)
 |
o “The two Colorado Creek, AK mammoths had radiocarbon ages of 22,850 +/- 670 and 16,150+/- 230 years respectively.” (Robert M. Thorson and R. Dale Guthrie, “Stratigraphy of the Colorado Creek Mammoth Locality, Alaska,” Quaternary Research, Vol. 37, No. 2, March 1992, pp. 214-228, see also: In the Beginning Walt Brown p. 124)
Problems With Carbon-14 Dating
The troubles of the radiocarbon dating method are undeniably deep and serious. Despite 35 years of technological refinement and better understanding, the underlying assumptions have been strongly challenged, and warnings are out that radiocarbon may soon find itself in a crisis situation. Continuing use of the method depends on a "fix-it-as-we-go" approach, allowing for contamination here, fractionation there, and calibration whenever possible. It should be no surprise, then, that fully half of the dates are rejected. The wonder is, surely, that the remaining half come to be accepted.
No matter how "useful" it is, though, the radiocarbon method is still not capable of yielding accurate and reliable results. There are gross discrepancies, the chronology is uneven and relative, and the accepted dates are actually selected dates.
Lee, Robert E. "Radiocarbon, Ages in Error, Anthropological Journal of Canada, Vol. 19, No. 3, 1981, pp. 9, 29 (Assistant Editor).
Apparent versus Actual Age
God did not make two babies in the Garden of Eden and give them seeds to plant. The creation was mature.
 |
• When God created the world, he created it with the appearance of age. For example:
– When Adam was first made he probably looked 30 to 40 years old (when in reality he was zero years old!)
– When God made the first trees, they probably looked 50 to 100 years old, maybe older. They might have even had tree rings!
– When God made the first rocks, they might have looked millions or perhaps billions of years old by today's "scientific" reckoning.
• Some people have a problem with the idea of God creating things with an apparent age because they think that it would be deceptive for God to do this. There are two responses to this objection:
– For God to make something instantaneously, He would have to give it an apparent age
– It is not deceptive, because God tells us what He did. If we fail to believe God, then we deceive ourselves!
Scientific “Proofs” for a Young Earth
 |
There are a number of scientific “proofs” that the earth is much younger than secular scientists now claim. Secular scientists generally reject such proofs because they do not give the “correct” results. Of course, like any other dating method (including the radioactive methods) these proofs are only as good as their assumptions. And it is on this basis that such proofs are often challenged - sometimes rightfully so. It is interesting to note that prior to the advent of radioactive dating methods, such methods were often used to estimate the age of the earth. Morris and Parker (What is Creation Science? Master Books, 1982, pp. 288-293) list 68 such proofs which, if we assume everything on the earth has always operated as it does today, yield a maximum age for the earth of anywhere from 140 years to 500,000,000 years!
|
1. |
Decay of earth's magnetic field |
10,000 years |
|
2. |
Influx of radiocarbon to the earth system |
10,000 years |
|
3. |
Influx of meteoritic dust from space |
too small to calculate |
|
4. |
Influx of juvenile water to oceans |
340,000,000 years |
|
5. |
Influx of magma from mantle to form crust |
500,000,000 years |
|
6. |
Growth of oldest living part of biosphere |
5,000 years |
|
7. |
Origin of human civilizations |
5,000 years |
|
8. |
Efflux of Helium-4 into the atmosphere |
1,750 - 175,000 years |
|
9. |
Development of total human population |
4,000 years |
|
10. |
Influx of sediment to the ocean via rivers |
30,000,000 years |
|
11. |
Erosion of sediment from continents |
14,000,000 years |
|
12. |
Leaching of sodium from continents |
32,000,000 years |
|
13. |
Leaching of chlorine from continents |
1,000,000 years |
|
14. |
Leaching of calcium from continents |
12,000,000 years |
|
15. |
Influx of carbonate to the ocean |
100,000 years |
|
16. |
Influx of sulphate to the ocean |
10,000,000 years |
|
17. |
Influx of chlorine to the ocean |
164,000,000 years |
|
18. |
Influx of calcium to the ocean |
1,000,000 years |
|
19. |
Influx of uranium to the ocean |
1,260,000 years |
|
20. |
Efflux of oil from traps by fluid pressure |
10,000 - 100,000 years |
|
21. |
Formation of radiogenic lead by neutron capture |
too small to measure |
|
22. |
Formation of radiogenic strontium by neutron capture |
too small to measure |
|
23. |
Decay of natural remanent paleomagnetism |
100,000 years |
|
24. |
Decay of C-14 in pre-Cambrian wood |
4,000 years |
|
25. |
Decay of uranium with initial "radiogenic" lead |
too small to measure |
|
26. |
Decay of potassium with entrapped argon |
too small to measure |
|
27. |
Formation of river deltas |
5,000 years |
|
28. |
Submarine oil seepage into oceans |
50,000,000 years |
|
29. |
Decay of natural plutonium |
80,000,000 years |
|
30. |
Decay of lines of galaxies |
10,000,000 years |
|
31. |
Expanding interstellar gas |
60,000,000 years |
|
32. |
Decay of short-period comets |
10,000 years |
|
33. |
Decay of long-period comets |
1,000,000 years |
|
34. |
Influx of small particles to the sun |
83,000 years |
|
35. |
Maximum life of meteor showers |
5,000,000 years |
|
36. |
Accumulation of dust on the moon |
200,000 years |
|
37. |
Instability of rings of Saturn |
1,000,000 years |
|
38. |
Escape of methane from Titan |
20,000,000 years |
|
39. |
Deceleration of earth by tidal friction |
500,000,000 years |
|
40. |
Cooling of the earth by heat efflux |
24,000,000 years |
|
41. |
Accumulation of calcareous ooze on sea floor |
5,000,000 years |
|
42. |
Influx of sodium to the ocean via rivers |
260,000,000 years |
|
43. |
Influx of nickel. to the ocean via rivers |
9,000 years |
|
44. |
Influx of magnesium to the ocean via rivers |
45,000,000 years |
|
45. |
Influx of silicon to the ocean via rivers |
8,000 years |
|
46. |
Influx of potassium to the ocean via rivers |
11,000,000 years |
|
47. |
Influx of copper to the ocean via rivers |
50,000 years |
|
48. |
Influx of gold to the ocean via rivers |
560,000 years |
|
49. |
Influx of silver to the ocean via rivers |
2,100,000 years |
|
50. |
Influx of mercury to the ocean via rivers |
42,000 years |
|
51 |
Influx of lead to the ocean via rivers |
2,000 years |
|
52. |
Influx of tin to the ocean via rivers |
100,000 years |
|
53. |
Influx of aluminum to the ocean via rivers |
100 years |
|
54. |
Influx of lithium into ocean via rivers |
20,000,000 years |
|
55. |
Influx of titanium into ocean via rivers |
160 years |
|
56. |
Influx of chromium into ocean via rivers |
350 years |
|
57. |
Influx of manganese into ocean via rivers |
1,400 years |
|
58. |
Influx of iron into ocean via rivers |
140 years |
|
59. |
Influx of cobalt into ocean via rivers |
18,000 years |
|
60. |
Influx of zinc into ocean via rivers |
180,000 years |
|
61. |
Influx of rubidium into ocean via rivers |
270,000 years |
|
62. |
Influx of strontium into ocean via rivers |
19,000,000 years |
|
63. |
Influx of bismuth into ocean via rivers |
45,000 years |
|
64. |
Influx of thorium into ocean via rivers |
350 years |
|
65. |
Influx of antimony into ocean via rivers |
350,000 years |
|
66. |
Influx of tungsten into ocean via rivers |
1,000 years |
|
67. |
Influx of barium into ocean via rivers |
84,000 years |
|
68. |
Influx of molybdenum into ocean via rivers |
500,000 years |